As the engine is operated, these residues gradually adhere to or are deposited in the exhaust gas system, and it is estimated that the deposits vary according to the combustion conditions, temperature, the pressure, gas flow speed, etc., in the system.
丂
1]丂Water content
Because a large volume of water is contained in the combustion product, as shown in the following formulae, it is, of course, believed that water is contained in the residue. However, in reality, the temperature in the exhaust gas piping system is high, and as a result the possibility that water is condensed and taken into the residue is low, and the water content of the residue is relatively low.
丂
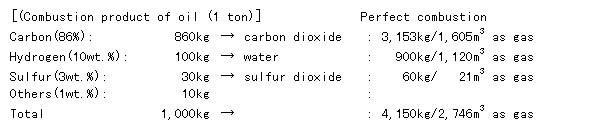
丂
2]丂Toluene solubles
The toluene solubles is the produced when the petroleum component in the fuel oil or the lubricating oil is not completely burned and discharged from the engine, i.e., the unburnt petroleum component. The unburnt oil part in the exhaust gas is essentially minimal if the combustion condition is excellent, and the existence (content) of the unburnt oil part in the residue reflects the combustion conditions (complete/incomplete) in the engine.
3]丂Toluene insolubles
The toluene insolubles forms about 80-90% of the residue, and consists of a combustible part (solid carbon) and ash (inorganic matter).
The amount of solid carbon becomes high toward the exhaust gas economizer on the downstream side of the exhaust system from the turbocharger immediately behind the combustion chamber, while the content of ash tends to be lower.
(2)丂PH
The "PH" of the combustion residue suspended in water indicates acidity (PH丗2-4) with most samples. (Fig. 1-1) On the other hand, from the results of the fluorescent X-ray and X-ray diffraction, it is confirmed that Calcium sulfide (CaSO4) is the major component of the inorganic material, and also that "SOx" in the combustion product is incorporated in the combustion residue. However, the major component in the residue from the generator engine is not calcium but iron. Sulfur in the heavy oil is burnt into sulfur dioxide (SO2), and a part of it is further oxidized and reacts with water to become corrosive sulfuric acid (strong acid)
丂
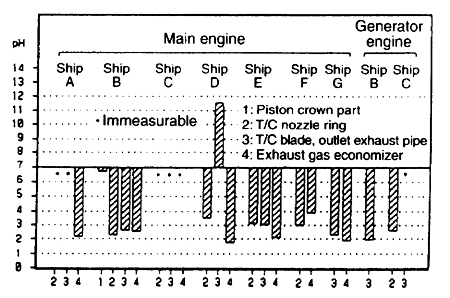
Fig. 1-1丂pH (combustion residue)
丂
The sulfuric acid adheres to the deposits in the exhaust gas pipes and the residue indicates acidity.
The sulfur oxide is neutralized with the LO additive (alkali) on the lubrication surface of the cylinder liner or the piston ring into calcium sulfide. However, not all the sulfur oxide in the exhaust gas is neutralized and most of it (SO2 or SO3) is discharged as exhaust gas.
However, part of the sample residue indicates neutrality or alkalinity. That is, two samples of B01 (PH=6.7) and D03 (PH=11.6) are higher in "PH" compared to other samples, and it is confirmed by X-ray that calcium carbonate (CaCO3) is contained in these samples in addition to calcium sulfide (CaSO4). Calcium carbonate is the additive in the cylinder oil which is neutralized with "SOx" and discharged. Also, because the alkali is always excessive on the lubrication surface of the cylinder liner, the residue in the engine is one of the causes of alkalinity.
Fig. 1-2 shows the plotted relationship between the content ratio (Ca/S ratio) of calcium to sulfur in the residue and PH of the residue. The PH of the residue with a higher calcium content compared with sulfur is high, proving the above-mentioned prediction.
Because the exhaust gas is acid gas containing SOx and NOx, the alkali additive discharged from the engines is gradually neutralized in the gas atmosphere even when the alkali additive is deposited.
丂
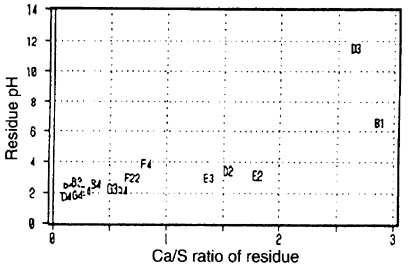
Fig. 1-2丂Relationship between pH and calcium
丂
丂
丂
BACK丂丂丂CONTENTS丂丂丂NEXT
丂